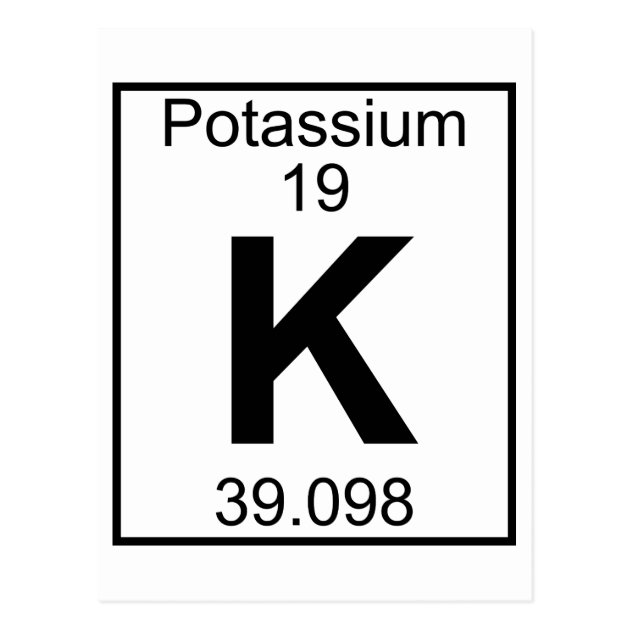
Potassium oxidises quickly in the presence of Oxygen so it must be stored in oil. Potassium is more reactive than Carbon on the reactivity series so it must be extracted from its ore using electrolysis. Properties Potassium is a more reactive alkali metal than Sodium but less reactive than Rubidium. Potassium ions have lost an electron to become positively charged. An atom of Potassium has only 1 electron in its outer shell. Atomic Structure The most stable isotope of Potassium has 20 neutrons in its nucleus giving it an atomic mass of 39. Potassium atoms join together in a giant metallic structure. Potassium is a Group 1 element, on the Periodic Table, with 19 protons in the nucleus.Ībout Potassium Molecular Structure Potassium has the chemical symbol K. Key Stage 4Ī 2 dimensional representation of the Bohr Model of a Potassium-39 isotope with 19 protons and 20 neutrons in the nucleus and 2 electrons in the first shell, 8 in the second, 8 in the third and 1 in the outer shell. Potassium is a solid at room temperature. Potassium reacts strongly with water to produce Hydrogen gas and Potassium Hydroxide. Atomic Structure Potassium has 19 protons and 20 neutrons in its nucleus giving it an Atomic Number of 19 and an atomic mass of 39. Potassium atoms join together in large numbers to form a giant metal molecule. Potassium is a Group 1 element, on the Periodic Table, with an atomic number of 19.Ībout Potassium Molecular Structure Potassium has the chemical symbol K. A 2 dimensional representation of a Potassium atom with 19 protons and 20 neutrons in the nucleus and 19 electrons orbiting the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
